da Leadership Medica n. 1 del 2001
ABSTRACT
 Chronic infection with hepatitis B virus and hepatitis C virus are major risk factors for hepatocellular carcinoma (HCC). HCC risk is particularly high in carriers with high liver cell proliferative activity and those with advanced liver disease. HCC often develops as a slowly growing single nodule. However, 40% of the patients have more than one HCC nodule when the tumor is first detected by imaging. Some tumors may rapidly grow. Screening with serum alphafetoprotein and abdominal ultrasound leads to identification of many patients with small, potentially operable tumors. However, it is not clear whether mortality from HCC is reduced in parallel. Treatment of HCC depends largely on the stage of the tumor and cirrhosis, but is limited overall by the lack of efficient chemotherapy. Transplantation offers the best chances of cure for small tumors. However, intention-to-treat reanalysis of the results yielded less encouraging figures than previously thought. Different locoregional treatment modalities may palliate/control the tumor disease in patients who are not eligible for operation. The 5-year survival of inoperable patients with a small HCC and compensated cirrhosis was approximately 50%. The substantial heterogeneity of survival between control groups makes it impossible to compare the results of individual trials.
Chronic infection with hepatitis B virus and hepatitis C virus are major risk factors for hepatocellular carcinoma (HCC). HCC risk is particularly high in carriers with high liver cell proliferative activity and those with advanced liver disease. HCC often develops as a slowly growing single nodule. However, 40% of the patients have more than one HCC nodule when the tumor is first detected by imaging. Some tumors may rapidly grow. Screening with serum alphafetoprotein and abdominal ultrasound leads to identification of many patients with small, potentially operable tumors. However, it is not clear whether mortality from HCC is reduced in parallel. Treatment of HCC depends largely on the stage of the tumor and cirrhosis, but is limited overall by the lack of efficient chemotherapy. Transplantation offers the best chances of cure for small tumors. However, intention-to-treat reanalysis of the results yielded less encouraging figures than previously thought. Different locoregional treatment modalities may palliate/control the tumor disease in patients who are not eligible for operation. The 5-year survival of inoperable patients with a small HCC and compensated cirrhosis was approximately 50%. The substantial heterogeneity of survival between control groups makes it impossible to compare the results of individual trials.
Introduction
The widespread adoption of ultrasound (US)-scan for screening high risk patients, has led to the identification of many patients with a small potentially treatable hepatocellular carcinoma (HCC) (1) (Figure 1 and 2). However, due to the lack of randomised controlled studies, it is not clear whether the mortality rate for HCC has been reduced in parallel. Such features of the natural history of HCC as the long-lasting subclinical incubation period of HCC that have been observed in many patients, as well as the large number of tumors which grow as a solitary mass seem to favour screening programs. Conversely, in many patients such aspects of HCC as multinodal onset of the tumor and great variations in the growth rates of single nodes, with doubling volume times from one to 20 months, may hinder the effectiveness of screening (2).

Fig. 1
Fig. 2
Population to be screened
As HCC is linked to environmental, dietary and life-style factors, epidemiological surveys were instrumental in identifying groups of individuals who are at risk for HCC. In two Consensus Conferences held in Anchorage (Alaska, USA) (3) and in Milan (Italy) (4) patients with cirrhosis, chronic carriers of hepatitis B surface antigen (HBsAg) and patients with rare metabolic liver diseases were identified as populations at risk for HCC. To improve cost-effectiveness of screening studies aimed at assessing individual’s cancer risk have been pursued. However, this is difficult because of individual variations in the metabolism of carcinogens, DNA repair capacities, genomic stability and inherited cancer predisposition.
Patients with cirrhosis
Four percent of all patients with cirrhosis due to chronic viral hepatitis or alcohol abuse develops HCC each year (1,5,6). HCC developed also in patients with chronic viral hepatitis without histologic evidence of cirrhosis, but at lower incidence rates. HCC developed in 110 Italian patients with cirrhosis out of a cohort of 417 followed since 1985. The annual rates of HCC development were 11% for the subgroup of men older than 53 yr with higher than 20ng/ml serum AFP levels. In a prospective study of 208 patients with compensated cirrhosis, HCC developed more often in patients with high liver cell proliferation than in those with low proliferation indices (annual rates: 5% vs 1%) (Figure 3). Thus, cirrhotic patients with exceptionally high risk of developing HCC can be identified to be enrolled in more cost-effective programs of HCC screening.
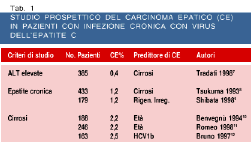
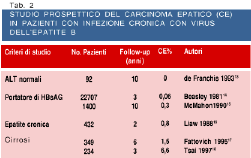
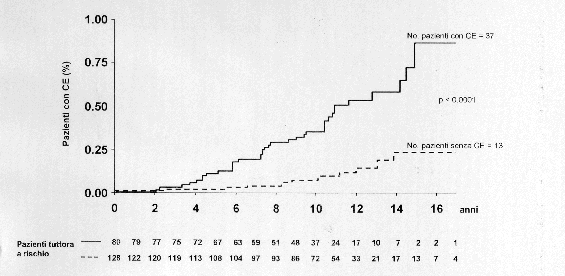
Fig. 3
Chronic carriers of HBsAg
More than 250 million persons are persistently infected with HBV, world-wide (19). Epidemiological, clinical and experimental studies have established a strong link between chronic infection with this virus and HCC. HBV is, in fact, responsible for both genotoxic lesions of the liver cells and tumor promotion through increased liver cell proliferation associated with persisting hepatitis and does not necessarily require the step to cirrhosis to be oncogenic (20). In a prospective cohort study in Taiwan, the risk of HCC in 3,454 HBsAg carriers was 102 times greater than in 19,253 non carriers (14). However, the carriers at especially high risk for developing HCC were those with actively replicating HBV (HBeAg+/HBV-DNA+) and those with cirrhosis. Healthy carriers may develop HCC, but are at substantially lower risk (13). Often distinguishing between clinical subsets of HBsAg carriers may be difficult, unless the patients’ are periodically assessed with laboratory or histological investigations. The strong link between HBV and HCC has been further confirmed by the decrease of HCC among Taiwanese children that has been observed since the start of mass vaccination of all newborns against HBV (21).
Patients with rare metabolic diseases
Patients with porphyria cutanea tarda, genetic hemochromatosis, alpha-1-anti-trypsin deficiency, tyrosinemia and hypercitrullinemia are also at high risk for HCC. Patients with glycogenosis type I and III, Wilson’s disease and hereditary fructose intolerance may also develop HCC, but are at substantially lower risk (2). HCC has developed also in patients with primary biliary cirrhosis, probably reflecting treatment-related improvement in patient’s survival. HCC was also found in 64% of 160 Japanese patients and in 48% of 101 South African Blacks with Budd-Chiari syndrome (22,23).
Screening tests
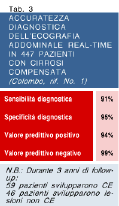
Screening tests must be simple, safe, acceptable to the patients and effective. Both serum alphafetoprotein (AFP) and abdominal ultrasound (US) fulfill these requisites, though they differ in terms of sensitivity and cost. The normal range for serum AFP is 0 to 20 ng/ml for the healthy adults. Serum levels of 400 ng/ml are very suggestive of HCC. However, two thirds of patients with small HCCs have less than 200 ng/ml and more than 30% of patients with HCC do not have abnormal circulating levels of AFP even in advanced stages (1,6,8). Moreover, between the range of 20 and 200 ng/ml, patients with chronic liver disease and false positive results due to hepatitis flares outnumber those with HCC. In patients with borderline elevations of serum AFP, the microheterogeneity of the sugar component of AFP can be assessed by lectin affinity electrophoresis coupled with antibody-affinity blotting. In a prospective study of 361 cirrhotics, 33 showed elevated atypical AFP 3 to 18 months before HCC was detected by imaging techniques (24). To minimise the false results of AFP determinations, cirrhotics are being surveilled by means of real-time US. As a general rule, a lesion seen as a discrete node in the liver should be presumed to be a preneoplastic lesion or HCC, and should be investigated accordingly. Most HCCs can be detected as a hypo-dysechoic mass (1,6,8).
Screening strategies
Screening strategies are adopted according to the local epidemiological and socioeconomic situation. In population-based studies, most individuals are asymptomatic, and therefore the screening intervals can be easily standardised. Serum AFP is the screening method of choice because these studies include thousands of persons and relatively few patients with liver disease at risk of false positive results with AFP. Because of the high risk of false results, AFP is not appropriate for screening cirrhotics. Abdominal US is the screening and surveillance method of choice. In fact, in 33-60% of the cirrhotics studied by US, HCC would have gone undetected if the patients had been screened by AFP only (1,6,24). The 6-month interval was thought to be the convenient screening interval for cirrhotic patients whose risk of developing HCC is approximately 30 times greater than that of healthy carriers of HBsAg.
Selection of patients to treat
Treatment options have been selected according to the presence or absence of cirrhosis, number and size of tumors, and degree of hepatic deterioration. Staging is a crucial variable in treatment outcome, since many therapeutic failures have resulted from incorrect patient selection. When patients are scanned by biphasic spiral computed tomography (CT) during the arterial phase highly vascularised tumors appear against a background of relatively unhenanced liver that is primarely enhanced during the late portal vein phase (25). In a multicenter study in Milan (26), 43% of 178 patients with early detected cancer had multinodular HCCs. However, most tumors smaller than two cm are hypovascular and therefore escape detection with such gold standard staging techniques as biphasic spiral-CT. For staging clinical status, the Child-Pugh scoring system provides accurate estimates of patient survival. The three year survival of untreated patients with a small tumor and well-compensated cirrhosis was approximately 25% (27).

Patients with normal livers
Hepatic resection is the primary option for patients with HCC who present with normal livers and well-preserved hepatic function. In two studies (28,29), the cumulative five-year survival for 128 such patients treated in 2 Centers with hepatic resection was approximately 45%.
Patients with cirrhosis and a small tumor
The functional capacity of the liver not involved by HCC is the major factor in these patients’ prognosis. Thus, hepatic resection was substantially successful in patients with small tumors and well preserved hepatic function. More recently, in the wake of substantial improvements in transplantation technology and better understanding of the natural history of HCC, liver transplantation has gained more popularity as the real curative treatment for patients with HCC and cirrhosis.
Orthotopic Liver Transplantation (OLT)
Liver transplant eliminates both detectable and undetectable tumor nodes and all the preneoplastic lesions in the cirrhotic liver. Removal of the diseased liver also reduces the risk of morbidity and mortality from portal hypertension. Opposing these “pros” for OLT are several important “cons”, for example, shortage of donated organs, high costs of the procedure, stringent criteria for selection of patients, high risk of early tumor recurrence due to faulty staging of the disease and immunosuppression, and recurrence of hepatitis. One major obstacle to the interpretation of OLT results are the large differences between transplantation centers in terms of time-lag between candidacy and operation. Between January 1988 and June 1994, the five-year survival of 537 patients with HCC (7% of total) who were given transplants in 82 European Centers was 39%. This included the 54.5% survival of 176 patients in whom cirrhosis was the primary indication for OLT and 45.5% of 361 cirrhotics in whom HCC was the primary indication. In Milan, 48 consecutive patients with viral cirrhosis and a single < 5 cm tumor or fewer than 3 < 3 cm nodes were treated by OLT (30). The 4-year actuarial survival was 92% for the 35 patients who were confirmed at the operation as having met the selection criteria, compared to 60% for the 13 patients who did not, because they were found to have ancillary nodes. Although survival of transplanted patients seems to be largely influenced by tumor size and number, there is no general agreement on the ideal tumor size that entails the least risk of recurrence, mostly because small tumor volume does not mean an early biological stage for all cases. Indeed, vascular invasion by the tumor and perihepatic lymph nodes can occur even in patients with small HCCs (28, 29). Unfortunately, vascular invasion can be assessed only during the operation and lymph nodes can be precisely assessed only during laparoscopy or laparotomy. The outcome of OLT may be influenced by recurrence of viral hepatitis, since infection of the graft may facilitate rejection and re-establish the oncogenic potential of the liver. The efficacy of interferon against hepatitis C is under evaluation. For hepatitis B, hyperimmune gamma-globulins and the nucleoside analogue Lamivudine are protective but costly (31,32).
Hepatic resection
Liver transplantation cannot be offered to all patients with cirrhosis who are found to harbour a small HCC. Thus, in many countries hepatic resection remains the primary therapeutic option for these patients. Since the functional capacity of the remaining liver is a major factor affecting prognosis for patients undergoing hepatic resection, limited hepatic resections (segmentectomy and subsegmentectomy) are the technical procedures of choice. Since 1983 the widespread adoption of intraoperative US has changed the outlook of this treatment. The best results in terms of both short-term and long-term survival were for patients with single tumors less than 2 cm in diameter and well preserved hepatic function. In 347 Japanese patients, the 5-year survival rate was as high as 60.5% (33), with very low mortality rates (0-5%). The 3-year cumulative survival was 50% for 78 Japanese patients with single tumors and Child’s A status, 35% for 26 with Child’s B status and 0 for three with Child’s C status (34). For 72 European patients, these figures were 51% for Child’s A patients and 12% for Child’s B-C (35). In patients with Child-Pugh A cirrhosis, portal hypertension is the most reliable predictor of survival after resection (Table 6).
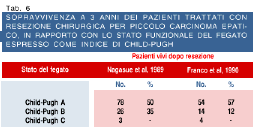
None of the 14 operated patients with less than 10 mmHg hepatic venous pressure gradient had had unresolved hepatic decompensation compared to 11 of the 15 patients with higher gradients (36). Thus, resection is definitely contraindicated for patients with deteriorated cirrhosis or severe portal hypertension in view of the high operative risk and short life expectancy. In Barcelona, the intention-to-treat analysis of patients treated by transplantation or resection demonstrated better survivals for resected patients with compensated liver and mild portal hypertension compared to similar patients who were transplanted in the last years when the waiting list rose from 62 days to 162 days, on average (37).
Patients with advanced disease
Patients could be refused surgery because of advanced age, deteriorated liver function, large tumors, tumors localised in strategic positions or associated clinical conditions which contraindicate surgery.
Percutaneous interstitial treatments
US-guided interstitial treatments include tumor injection with absolute ethanol, 50% acetic acid or hot saline, or tumor thermoablation with radiofrequency, microwaves or laser. Most treatments were carried out with intratumor ethanol injection (PEI), which causes extensive coagulative necrosis of the tumor cells, and thrombosis of the tumor vessels, and is well tolerated (Figure 4 and 5).

Fig. 4
Fig. 5
Once more, survival was largely influenced by liver function, size and number of tumors. The 5-year survival of 293 Italian patients with Child’s A cirrhosis and a small HCC was 47%, compared to 29% for 149 with Child’s B cirrhosis (38) (Table 7).

Life expectancy of Child’s A patients with a small tumor treated by PEI appeared to be as good as that of similar patients treated with hepatic resection, and associated with a low risk of severe complications (1.7%) and mortality (0.1%). In 60 randomly selected patients with tumors smaller than 3 cm and compensated cirrhosis, injection with 50% acetic acid was superior to ethanol in terms of 2-year cancer-free survival (92% vs 63%, P=0.02), being particularly active in patients with hypervascular tumors (39). Radiofrequency thermoablation is safe and convenient in patients with compensated cirrhosis and a small HCC. In a prospective randomised study of 86 patients with compensated cirrhosis and small HCC, radiofrequency was superior to PEI in terms of complete tumor necrosis (90% vs 80%), and numbers of treatments (1.2 vs 4.8), but it caused more complications (9.5% vs 0) (40).
Transcatheter Arterial Chemo-Embolization (TACE)
Transcatheter arterial embolization of HCC is possible because the liver has a dual blood supply while HCC is supplied virtually only from the hepatic artery. TACE through the femoral artery leads to ischemic necrosis of the tumor and makes hepatic arterial injection of antitumor agents possible, giving higher local concentrations of drugs with fewer systemic side effects. TACE of the proximal hepatic artery (conventional TACE) has been widely employed in Eastern and Western countries as an alternative to hepatic resection and has now been improved, as segmental or subsegmental TACE. The procedure is contraindicated for patients with venous tumor supply (hypovascular tumors), advanced liver deterioration, complete thrombosis of the portal vein trunk, renal failure or extrahepatic metastases. In the last decade, three randomised controlled studies of TACE and one randomised controlled study of transarterial embolization (TAE, without chemotherapy) treatment of patients with unresectable HCC have been conducted (41-44) Conversely, non-controlled studies of segmental TACE in 63 Japanese patients with Child-Pugh A cirrhosis and small HCC reported four-year survivals comparable to those for similar patients treated with resection or PEI (45). Thus, TACE needs to be reassessed for treating patients with compensated cirrhosis and a small vascularised HCC.
Other treatments
Systemic chemotherapy has been widely used to treat inoperable HCC, but the response rate is very low (20%) (46). The possible sex hormone-dependence of HCC and the presence of tumor hormone receptors have suggested a potential for hormonal manipulation of tumor growth, particularly using anti-estrogens. However, in two large studies of 120 and 477 patients with inoperable HCC, but less deteriorated liver functions, treatment with the anti-estrogen tamoxifen did not improve survival or the quality of life, compared to controls (47,48). In a prospective randomised study of 58 patients with advanced HCC, treatment with subcutaneous octreotide 250 mcg twice daily significantly improved survival at 12 months (49). However, the sample size of the study was not adequate for preventing a type II error in the assessment of the difference between treatment and no-treatment. The conventional method of external irradiation is not effective against HCC. Using three-dimensional radiation planning (conformal radiotherapy) the beam scatter can be minimised to deliver the therapeutic dose, making selective irradiation of the liver possible. Local radiation was performed safely in patients with Child A cirrhosis and smaller than 8 cm tumor, with a partial response rate of 64% (50). With proton radiation therapy, a large amount of radiation is focused only on the lesion, and the exposure of surrounding non-tumoral liver can be limited. Of 83 patients so treated only 19% had a complete response without any appreciable effect on survival. In the near future, the gene therapy may offer the chance to cure many patients suffering from Hepatocarcinoma. The principle consists in transfecting the neoplastic cells with a virus capable of introducing a gene which facilitates the suicide of the cell, or therapeutic genes which can make the neoplastic hepatocytes responsive to antiviral drugs.
Conlusions
Early diagnosis of Hepatocarcinoma is possible, but we do not know whether screening reduces the mortality rate for this tumour, in that we lack checked clinical research works. Without doubt, screening can offer advantages, such as an improvement in the therapeutic response by patients who have benefited from an early diagnosis, for some of whom radical treatments can be applied, as well as the reassurance of patients with negative diagnostic tests. All this involves a saving in resources. However, screening can also involve disadvantages, such as inflicting a longer “morbility” on patients with a prognosis which cannot be influenced by treatment, giving false reassurance to patients with untruly negative results and unnecessary morbility to patients with untruly positive results. These disadvantages involve an increase in costs. Patients with cirrhosis or congenital metabolic diseases involving a risk of Hepatocarcinoma need to be checked more frequently (at six-month intervals) than HBsAg carriers with no signs of hepatic illness (at twelve-month intervals). Surgical resection is the selected treatment in the few patients suffering from tumour in a “healthy” liver. Orthotopic liver transplantation continues to be the best solution for patients with cirrhosis and a tumour smaller than 5 cm in size. However, owing to the lack in organ donors, many centres continue to regard surgical resection as the first “therapeutic” option. Palliative treatments, such as percutaneous injection of alcohol and radiofrequency, are indicated for patients with a well-balanced liver suffering from a neoplastic disease which cannot be surgically treated. In many patients, the most effective approach is given by a combined resort to these treatments, that is targeted ablation therapies and resectional surgery or transplantation.
Massimo Colombo
Direttore, Cattedra di Medicina Interna, Centro “A.M. e A. Migliavacca” per lo Studio del Fegato
IRCCS Ospedale Maggiore, Milano
Bibliography
- Colombo M, de Franchis R, Del Ninno E, Sangiovanni A, De Fazio C, Tommasini M, Donato MF, Piva A, Di Carlo V, Dioguardi N. Hepatocellular carcinoma in Italian patients with cirrhosis. N Engl J Med 1991;325:675-80.
- Colombo M. Hepatocellular carcinoma in cirrhotics. Semin Liv Dis 1993;13:374-83.
- Mc Mahon BJ, London T. J Natl Cancer Inst Workshop on screening for hepatocellular carcinoma. 1991;83:916-9.
- Colombo M. Early diagnosis of hepatocellular carcinoma in Italy. A summary of a Consensus Development Conference held in Milan, 16 November 1990 by the Italian Association for the Study of the Liver (AISF). J Hepatol 1992;14:401-3.
- Oka H, Kurioka N, Kim K, Kanno T, Kuroki T, Mizoguchi Y, Kobayashi K. Prospective study of early detection of hepatocellular carcinoma in patients with cirrhosis. Hepatology 1990;12:680-687.
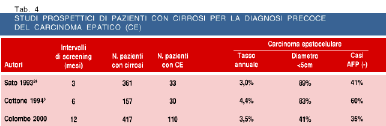
- Cottone M, Turri M, Caltagirone M, Parisi P, Orlando A, Fiorentino G, Virdone R, Fusco G, Grasso R, Simonetti RG, Pagliaro L. Screening for hepatocellular carcinoma in patients with Child’s A cirrhosis: a 8 year prospective study by Ultrasound and Alphafetoprotein. J Hepatol 1994;21:1029-1034.
- Tradati F, Colombo M, Mannucci PM, Rumi MG, De Fazio C, Gamba C, Ciavarella N, Rocino A, Morfini M, Scaraggi A, Taioli E, and The Study Group of the Association of Italian Hemophilia Centers. A prospective multicenter study of hepatocellular carcinoma in Italian hemophiliacs with chronic hepatitis C. Blood 1998;91:1173-7.
- Tsukuma H, Hiyama T, Tanaka S, Nakao M, Yabuuchi T, Kitamura T, Nakanishi K, Fujimoto I, Inoue A, Yamazaki H, Kawashima T. Risk factors for hepatocellular carcinoma among patients with chronic liver disease. N Engl J Med 1993;328:1797-801
- Shibata M, Morizane T, Uchida T, Yamagami T, Onozuka Y, Nakano M, Mitamura K, Ueno Y. Irregular regeneration of hepatocytes and risk of hepatocellular carcinoma in chronic hepatitis and cirrhosis with hepatitis-C-virus infection. Lancet 1998;351:1773-7.
- Benvegnù L, Fattovich G, Noventa V, Tremolada F, Chemello L, Cecchetto A, Alberti A. Concurrent hepatitis B and C virus infection and risk of hepatocellular carcinoma in cirrhosis: A prospective study. Cancer 1994;74:2442-8.
- Romeo R, Rumi MG, Del Ninno E, Colombo M. Hepatitis C virus genotype 1b and risk of hepatocellular carcinoma. Hepatology 1997;26:1077.
- Bruno S, Silini E, Crosignani A, Bonino F, Leandro G, Bono F, Asti M, Rossi S, Larghi A, Cerino A, Podda M, Mondelli MU. Hepatitis C virus genotypes and risk of hepatocellular carcinoma in cirrhosis: a prospective study. Hepatology 1997;25:754-8.
- de Franchis R, Meucci G, Vecchi M, Tatarella M, Colombo M, Del Ninno E, Rumi MG, Donato MF, Ronchi G. The natural history of asymptomatic hepatitis B surface antigen carriers. Ann Intern Med 1993;118:191-4.
- Beasley RP, Hwang LY, Lin CC, Chien CS. Hepatocellular carcinoma and hepatitis B virus. A prospective study of 22707 men in Taiwan. Lancet 1981;ii:1129-32.
- Mc Mahon BJ, Alberts SR, Wainwright RB, Bulkow L, Lanier AP. Hepatitis B sequelae: Prospective study in 1400 hepatitis B surface antigen-positive Alaska native carriers. Arch Intern Med 1990;150:1051-4.
- Liaw YF, Tai D, Chu CM, Lin DY, Sheen IS, Chen TJ, Pao CC. Early detection of hepatocellular carcinoma in patients with chronic type B hepatitis. A prospective study. Gastroenterology 1986;90:263-7.
- Fattovich G, Gustina G, Schalm SW, Hadziyannis S, Sanchez-Tapias J, Almasio P, Christensen E, Krosgaard K, Degos F, Carneiro de Moura M, Solinas A, Noventa F, Realdi G, The EUROHEP Study Group on Hepatitis B Virus and Cirrhosis. Occurrence of hepatocellular carcinoma and decompensation in Western European patients with cirrhosis type B. Hepatology 1995;21:77-8.
- Tsai JF, Jeng JE, Ho MS, Chang WY, Hsieh MY, Lin ZY, Tsai JH. Effect of hepatitis C and B virus infection on risk of hepatocellular carcinoma: a prospective study. Br J Cancer 1997;76:968-74.
- Maynard JE. Hepatitis B: Global importance and need for control. Vaccine 1990;8:S18-20
- Rogler CE. Cellular and molecular mechanisms of hepatocarcinogenesis associated with hepadnavirus infection. In: Mason WS, Seager C, eds. Hepadnavirus molecular biology and pathogenesis. Berlin: Springer-Verlag, 1991;102-40.
- Chang MH, Chen CJ, Lai MS, Hsu HM, Wu TC, Kong MS, Liang DC, Shau WY, Chen DS, for the Taiwan Childhood Hepatoma Study Group. Universal hepatitis B vaccination in Taiwan and the incidence of hepatocellular carcinoma in children. N Engl J Med 1997;336:1855-9.
- Nakamura T, Nakamura S, Aikawa T, Suzuki O, Onodera A, Karoji N. Obstruction of the inferior vena cava in the hepatic portion and the hepatic vein: Report of eight cases and review of the Japanese literature. Angiology 1968;19:479-98.
- Kew MC, McKnight A, Hodkingson H, Bukofzer S, Esser JD. The role of membranous obstruction of the inferior vena cava in the etiology of hepatocellular carcinoma in Southern African Blacks. Hepatology 1989;9:121-5.
- Sato Y, Nakata K, Kato Y, Shima M, Ishii N, Koji T, Taketa K, Endo Y, Nagataki S. Early recognition of hepatocellular carcinoma based on altered profiles of alpha-fetoprotein. N Engl J Med 1993;328:1802-6.
- Ros PR, Davis GL. The incidental focal liver lesion: Photon, proton or needle? Hepatology 1998;27:1183-90.
- Fasani P, Sangiovanni A, De Fazio C, Borzio M, Bruno S, Del Ninno E, Colombo M. High prevalence of multinodular hepatocellular carcinoma in patients with cirrhosis due to multiple etiological factors. Hepatology 1999;29:1704-7.
- Livraghi T, Bolondi L, Buscarini L, Cottone M, Mazziotti A, Morabito A, Torzilli G. No treatment, resection and ethanol injection in hepatocellular carcinoma: a retrospective analysis of survival in 391 patients with cirrhosis. J Hepatol 1995;22:522-6.
- Ringe B, Pichlmayr R, Wittekind C, Tush G. Surgical treatment of hepatocellular carcinoma: Experience with liver resection and transplantation in 198 patients. World J Surg 1991;15:270-85.
- Iwatsuki S, Starzl TE, Sheahan DG, Yokoyama I, Demetris AJ, Todo S, Tzakis AG, Van Thiel DH, Carr B, Selby R, Madariaga J. Hepatic resection versus transplantation for hepatocellular carcinoma. Ann Surgery 1991;214:221-9.
- Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F, Montalto F, Ammatuna M, Morabito A, Gennari L. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 1996;334:693-9.
- Bain VG, Kneteman NM, Ma MM, Gutfreund K. Shapiro JA, Fischer K, Tipples G, Lee H, Jewell LD, Tyrrell DL. Efficacy of Lamivudine in chronic hepatitis B patients with active viral replication and decompensated cirrhosis undergoing liver transplantation. Transplantation 1996;62:1456-62.
- Samuel D, Muller R, Alexander G, Fassati L, Ducot B, Benhamou JP, Bismuth H. Liver transplantation in European patients with the hepatitis B surface antigen. N Engl J Med 1993;329:1842-7.
- Tobe T, Arii S. Improving survival after resection of hepatocellular carcinoma: Characteristics and current status of surgical treatment of primary liver cancer in Japan. In: Tobe T, Kameda H, Okudaira M, Otho M, Endo Y, Mito M, Okamoto E, Tanikawa K, Kojiro M, eds. Primary Liver Cancer in Japan. Tokyo, Berlin, Heidelberg, New York, London, Paris, Hong Kong, Barcelona, Springer 1992;215-20.
- Nagasue N, Yukaya H. Liver resection for hepatocellular carcinoma: Results from 150 consecutive patients. Cancer Chemother Pharmacol 1989;23:S78-82.
- Franco D, Capussotti L, Smadja C, Bouzari H, Meakins J, Kemeny F, Grange D, Dellepiane M. Resection of hepatocellular carcinomas. Results in 72 European patients with cirrhosis. Gastroenterology 1990;98:733-8.
- Bruix J, Castells A, Bosch J, Feu F, Fuster J, Garcia-Pagan JC, Visa J, Bru C, Rodes J. Surgical resection of hepatocellular carcinoma in cirrhotic patients: prognostic value of preoperative portal pressure. Gastroenterology 1996;111:1018-1023.
- Llovet JM, Fuster J, Bruix J, for the Barcelona Clinic Liver Cancer (BCLC) Group. Intention-to-treat analysis of surgical treatment for early hepatocellular carcinoma: resection versus transplantation. Hepatology 1999;30:1434-40.
- Livraghi T, Giorgio T, Marin G, Salmi A, de Sio I, Bolondi L, Pompili M, Brunello F, Lazzaroni S, Torzilli G, Zucchi A. Hepatocellular carcinoma in cirrhosis in 746 patients: Long-term results of percutaneous ethanol injection. Radiology 1995;197:101-8.
- Ohnishi K, Yoshioka H, Ito S, Fujiwara K. Prospective randomized controlled trial comparing percutaneous acetic acid injection and percutaneous ethanol injection for small hepatocellular carcinoma. Hepatology 1998;27:67-72.
- Livraghi T, Goldberg SN, Lazzaroni S, Meloni F, Solbiati L, Gazelle GS. Small hepatocellular carcinoma: Treatment with radio-frequency ablation versus ethanol injection. Radiology 1999;210:655-61.
- Pelletier G, Roche A, Ink O, Anciaux ML, Derhy S, Rougier P, Lenoir C, Attali P, Etienne JP. A randomized trial of hepatic arterial chemoembolization in patients with unresectable hepatocellular carcinoma. J Hepatol 1990;11:181-4.
- Groupe d’Etude et de Traitment du Carcinome Hépatocellulaire. A comparison of Lipiodol chemoembolization and conservative treatment for unresectable hepatocellular carcinoma. N Engl J Med 1995;332:1256-61.
- Pelletier G, Ducreux M, Gay F, Luboinski M, Hagege H, Dao T, Van Steenbergen W, Buffet C, Rougier P, Adler M, Pignon JP, Roche A. Treatment of unresectable hepatocellular carcinoma with lipiodol chemoembolization: a multicenter randomized trial. J Hepatol 1998;28:129-34.
- Bruix J, Llovet JM, Castells A, Montana X, Bru C, Ayuso MC, Vilana R, Rodes J. Transarterial embolization versus symptomatic treatment in patients with advanced hepatocellular carcinoma: Results of a randomized controlled trial in a single institution. Hepatology 1998;27:1578-83.
- Matsui O, Kodoya M, Yoshikawa J, Gabata T, Arai K, Demachi H, Miyayama S, Takashima T, Unoura M, Kogayashi K. Small hepatocellular carcinoma: Treatment with subsegmental transcatheter arterial embolization. Radiology 1993;188:79-83.
- Lai CL, Wu PC, Chan GCB, Lok ASF, Lin HJ. Doxorubicin versus no antitumor therapy in inoperable hepatocellular carcinoma. A prospective randomized trial. Cancer 1988;62:479-83.
- Castells A, Bruix J, Bru C, Ayuso C, Roca M, Boix L, Vilana R, Rodes J. Treatment of hepatocellular carcinoma with tamoxifen: a double-blind placebo-controlled trial in 120 patients. Gastroenterology 1995;109:917-22.
- CLIP Group. Tamoxifen in treatment of hepatocellular carcinoma: a randomised controlled trial. Lancet 1998;352:17-20.
- Kouroumalis E, Skordilis P, Thermos K, Vasilaki A, Moshandrea J, Monousos ON. Treatment of hepatocellular carcinoma with octreotide: a randomised controlled study. Gut 1998;42:442-7.
- Lawrence TS, Tesser RJ, Tam Haken RK. An application of dose volume histograms to the treatment of intrahepatic malignancies with radiation therapy. Int J Radiat Oncol Biol Phys 1991;20:555-61.



