from Leadership Medica n. 9/2004
Abstract
Whipple’s disease is a rare systemic disease, whose first descriptions date back to the early 20th century. Tropheryma whippelii, the condition’s aetiological agent, is a Gram+ actinomycete, ubiquitous in the environment and frequently detected also in healthy individuals. Subsequently, the condition’s rare nature is related to the existence of an individual cofactor, most likely the deficit of interleukin 12, which causes the tendency to develop the disease. Clinically, Whipple’s disease appears extremely polymorphous, as it can cause the appearance of gastroenterological (malabsorption), cardiological, rheumatological, neurological and haematological syndromes mainly in males aged over 50 years. In particular, symptoms involving articulations appear years before the onset of intestinal ones. The neurological symptoms, which are the worst, are a prerogative especially of recurrences, which are secondary to the suspension of antibiotic treatment. This treatment should instead be administered constantly and today’s choice antibiotic is trimetoprim-sulfamethoxazole. To date the diagnosis is based on the presence of special alterations in intestinal biopsies, such as the presence of lamina propria macrophages with extensive frothy cytoplasm full of PAS+ material. Since it was first described Whipple’s disease was considered an intestinal disease with an uncertain aetiology, characterized by the presence in the small intestine’s lamina propria of cells with a frothy appearance, which are clinically responsible for a relevant malabsorption syndrome. (1) We shall see further on how the latest findings have modified and especially extended our knowledge on this condition, which, however has remained unchanged at least concerning one feature, its ascertained rarity.
Rare disease (all diseases with a prevalence below 5 cases / 10,000 individuals) does not absolutely mean unimportant disease. There are over 5,000 rare diseases. They can be fatal or however chronic and disabling, and they represent 10% of known diseases. Considering the limited market, the interest of pharmaceutical firms in developing research and in producing the so-called “orphan” drugs that are potentially useful for these pathologies is very poor, while instead rare diseases are a relevant social problem due to the difficulties patients often meet in obtaining adequate diagnoses and treatment. For these reasons too the Ministry of Health has created a national network to promote information about them, to develop preventive actions, to activate surveillance and to improve interventions focused on their diagnosis and treatment. The network has been formed by hospitals accredited for their clinical experience and scientific competence, furnished with supportive and complementary structures, including emergency services and biochemical, genetic and molecular diagnostics.
The Chair and Division of Gastroenterology of the Policlinico San Matteo, University of Pavia, is the national landmark for Whipple’s disease (2) and it belongs to an international research project on this condition sponsored by the European Community. (3)
A historical outline
In 1907, while still a student at the John’s Hopkins University, George Hoyt Whipple, Nobel Prize winner for Medicine for his studies on pernicious anaemia, described the case of a patient who died of a disease that was unknown at the time, characterized by large cell infiltration of intestinal mucous tissue and by mesenteric adenomegaly, pleuritis, pericarditis and endocarditis. (1) But this is not the “first” case of Whipple’s disease reported in literature. In 1895 Allchin and Hebb at the Westminster Hospital, London, described a patient with “gastric lymphangiectasia”, diarrhoea and vomiting, who died in a few months with a picture of acute malnutrition. (4) Many years later, in 1961, while reorganizing the Westminster Hospital museum’s historical archive Morgan, a pathologist, understood that Allchin and Hebb’s patient’s tissue was not stomach but tangentially sectioned small intestine, and he tried staining it with PAS, suspecting it was Whipple’s disease. (5) The preparation was intensely PAS positive, though 66 years had passed, confirming the diagnosis according to current criteria for this condition’s recognition.
Only another 14 cases were described from 1907 to 1943. Reports later increased in number due to the widespread use of intestinal biopsies: 12 new cases were reported till 1949 and 77 between 1950 and 1959. (6) The first diagnosis on a living patient dates back to 1947 and it was made by studying a lymph node, which had been laparotomically removed, thus suggesting that the disease could be located even in districts other than the small intestine. (7) The fact that in practice Whipple’s disease was a systemic disease (which, on the other hand, Whipple had anticipated in his first description) was cleared around ten years later with the description of patients who presented lesions similar to intestinal ones even in lymph nodes, articulations, endocardium, lungs and in the central nervous system. (8,9)
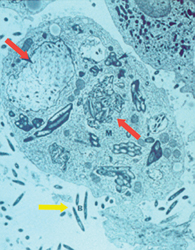
In the pathological context, in 1949 Black-Shaffer provided the histological pathognomic criterion to diagnose Whipple’s disease, in other words the presence of glycoproteins positive to staining with periodic acid-Schiff (PAS) in cells of the intestine’s lamina propria, which proved to be macrophages. (10) The first founded theories of the condition’s aetiology resulted from evidence that the use of broad-spectrum antibiotics, chloramphenicol or tetracyclines was followed by a ready, long-term clinical and, at least partly, histological, remission (11). The condition’s microbiological nature was confirmed by electron microscopic evidence of intestinal lesions (Figure 1), which enabled to ascertain that PAS+ material present in lysosomal vacuoles of macrophages (phagosomes) was, in practice, formed by Gram+ bacilli capsules in various stages of digestion. The latter were relatively monomorphous and 1.2 m x 0.25 m (12,13) in dimension. The fact that these micro-organisms had a low virulence was confirmed by the constant absence of tissue necrosis.
|
|
|
Figure 1 Ultrastructural appearance of a macrophage in intestinal mucous tissue in Whipple's disease. Red arrows point out phagosomes containing bacilli in various stages of digestion, while blue arrows point out bacilli in intercellular space. |
However Whipple’s disease’s aetiology was a mystery and remained thus for yet many years. Few, due to the response to antibiotics and the extensive presence of bacilli in the site of lesions, doubted the condition’s bacterial aetiology, which, however, did not form a real infectious disease due to the absence of evidence to back direct transmission, family cases or epidemic foci.
In 1992, at the University of Stanford, Relman and colleagues (14) applied the PCR method to amplify the nucleotidic sequence of the bacterial rRNA’s subunit 16 extracted from the intestinal biopsy of a patient suffering from Whipple’s disease. This enabled to identify a special 284-base sequence. The new bacillus thus identified was called Tropheryma whippelii and it was nosographically numbered among Gram+ Actinomycetes rich in guanine and cytosine, belonging to the Actinobacteria family. (15)
Table 1 summarises the main stages in the history of Whipple’s disease.
Epidemiology and Pathogenesis
Clinical symptoms, which are so complex and polymorphous, and aetiological factors, which have only recently been cleared, have not been this condition’s only attractive factors in the course of the years. Many obscure aspects have been solved with the identification of Tropheryma whippelii, but if the disease’s greater prevalence in the 5th and 6th decades of life can be explained by the time required for a bacillus with low virulence to cause the disease’s typical lesions, the virtually absolute incidence of Whipple’s disease in males (male/female ratio equal to 9/1) has yet to be explained. Table 2 shows the demographic and clinical features of a series of 14 patients followed by us. All our cases are male and only one, the most recent, was diagnosed before the 5th decade.
Soil is the main “reservoir” of Actinomycetes and a particularly interesting aspect of Whipple’s disease’s epidemiology is the fact that most patients reported in literature (and this coincides with our personal experience) were farmers, carpenters and miners or their main hobby was gardening. (6) Axel Von Herbay’s group (16) found the bacterium in 25 on 38 Rhine valley sullage water samples, thus confirming the contribution given by the discovery of Tropheryma whippelii to solving these issues. Hence Tropheryma is much more ubiquitous than one could reasonably expect. Besides the PCR method was applied to isolate it in 5% of intestinal biopsies and in 11% of gastric juice samples taken from 105 dyspeptic patients (17) and, even, in 35% of saliva samples from healthy individuals. (18) Since these tests’ specificity is adequately high, we could conclude that the presence of Tropheryma whippelii in the environment and in man is much greater than the incidence of Whipple’s disease; hence this suggests that the disease’s development requires individual factors, which can influence the transition from “infection” to disease and which determine the latter’s rarity.
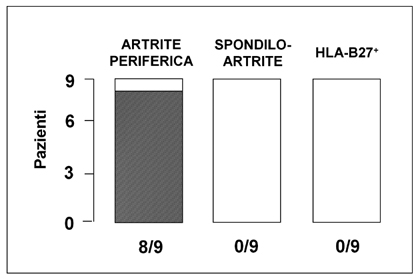
Though Whipple’s disease is a condition that presents many immunological repercussions, its predisposition does not seem related to the presence of genes belonging to the HLA system. A metanalysis performed by Dobbins (6) suggested a weak but significant association with HLA B27, which is extremely interesting if we think that HLA-B27 transgenic rats are carriers of chronic intestinal phlogosis and arthritis. Results achieved later both by our team (20) and by others (21) however confuted this association. In particular, none of the 9 patients studied by us was a carrier of spondylarthritis and nobody was HLA-B27+ (Fig. 2).
It is likely that an individual tendency to develop Whipple’s disease can be traced back to a defect in the immune system and, in particular, to the failure of the monocyte-macrophagic system’s cells to produce interleukin 12 (22,23). This deficiency could reduce the body’s defensive capacities towards intracellular bacteria (22), subsequently reducing macrophages’ capacity to remove cells infected by Tropheryma whippelii. In this regard, our team is evaluating possible alterations in IL-12 encoding and “promoter” genes.
 |
|
Figure 2 Prevalence of HLA-B27 vs. the rheumatological picture in 9 Italian patients suffering from Whipple's disease.(20) |
Clinical symptoms and complications
Table 3 reports the prevalence range of clinical symptoms in Whipple’s disease, which can be deduced from the critical review of some extensive series of patients reported in literature. It is clear, as already mentioned, that Whipple’s disease must not be considered an exclusively intestinal disease, but rather a systemic infection characterized by the accumulation of PAS+ material (in other words by the accumulation of bacteria) in various organs and systems.
Its clinical polymorphism, which can appear with gastroenterological (malabsorption), cardiological (endocarditis), rheumatological (seronegative migrating polyarthritis), neurological (ataxia, pyramidal and extrapyramidal disorders, progressive dementia, oculo-masticatory myorhythmia depending on the infection’s localization in the central nervous system) and haematological (thrombotic events related to reactive thrombocytosis or to other defects in coagulation) symptoms, essentially depends on this. (24,25)
But all clinical studies published so far are inclined to a basic “prejudice”. In fact their choice diagnostic investigation has always been the intestinal biopsy and this can have fictitiously increased the prevalence (near absolute values) of gastroenteric symptoms. Nothing prevents one from thinking that when the diagnosis will be based, in routine practice too, on applying molecular biology techniques to seek Tropheryma in other tissue and in other risk conditions (seronegative migrating arthritis, critical encephalitis, recurrent pleuropericarditis), its intestinal localization will not be more frequent and the disease’s global prevalence will increase. This has been recently confirmed by reports on forms of Tropheryma that do not involve the gastrointestinal system (26,27); on the other hand, the involvement of articulations generally precedes intestinal symptoms by a few years in over 60% of cases. (26,28)
Concerning these articular symptoms, sacroiliitis is axially far more frequent than ankylosing spondylitis. Due to the presence of arthritis and malabsorption, Whipple’s disease should be a condition that often causes osteoporosis. In practice a study conducted by us (29) shows that bone mineral density is significantly reduced only at the femur in patients suffering from Whipple’s disease, compared to control groups that are comparable in sex and age. Even metabolism and bone mineral turnover parameters are less compromised than in other enteropathies. Most likely in this condition the strong prevalence of males and the disease’s very onset at an age long past the moment peak bone mass is reached are protective factors towards the onset of osteoporosis.
Lastly, many of the disease’s clinical symptoms can be caused by genotypical and phenotypical differences between the various strains of Tropheryma whippelii. These differences vary the bacilli’s ability to involve different organs and systems. Two different bacterial strains called Slow-Marseille and Twist-Marseille, and 7 different variants on the basis of genotypical differences identified as Hinrikson’s type 1-3 (30,31), Maiwald’s type 4-6 (32) and Geissdorfer’s type 7 (33) have been currently recognized. However, the ratio between bacterial genotypes and clinical symptoms is still an unsolved problem: it has still to be ascertained whether bacteria found in individuals free of symptoms are related to the presence of non pathogenic forms (34), just as we still do not know whether there is any difference, in biomolecular terms, between bacteria isolated in patients with a classically systemic form and bacteria isolated from patients with atypical forms, which present few symptoms that are only endocardiac (35), rheumatological (36) or neurological. (37,38)
This disease’s recurrences, generally caused by the choice of antibiotic that cannot cross the haematoencephalic barrier or by the prolonged suspension of treatment, take on particularly serious features because they are often related to lesion localization in the central nervous system.
Diagnosis and Treatment
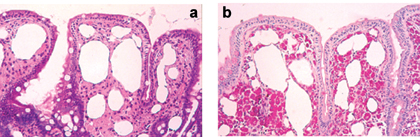
As already stated, the diagnosis is based on the intestinal biopsy. Figures 3(a) and 3(b) show typical alterations that are clearly noticeable in the intestinal mucous tissue of our patient suffering from Whipple’s disease. In (a) the presence in the lamina propria of cells with extensive frothy cytoplasm can be clearly noticed, stained with haematoxylin and eosine; (b) reveals how, following PAS staining, these cells (macrophages) are filled with red PAS+ material. These PAS+ macrophages’ massive infiltration of the lamina propria causes the other two typical histopathological alterations: compression of the villus’ lacteal, which hence appears dilated, and loss of the same villus’ normal structure, which becomes shorter and stubby.
 |
|
Figura 3 Appearance under light microscopy of intestinal biopsy, stained with haematoxylin-eosine (a) and periodic acid-Schiff (b), of patient suffering from Whipple's disease. |
Concerning treatment, Whipple’s disease can be fatal when left untreated. In most patients antibiotic treatment leads to a rapid improvement of clinical conditions and to a long-term remission. (6,39,40) The many therapeutic patterns so far used are exclusively based on empirical observations (40) and they have long failed to guarantee coverage towards the disease’s neurological recurrences. (41)
In practice tetracyclines and penicillins do not pass the haematoencephalic barrier and they do not prevent neurological complications; hence their single use must be avoided. Currently the choice antibiotic is trimetoprim-sulfamethoxazole (39), which must be administered more or less indefinitely. Its action is based, at least partly, on the competitive inhibition of the enzyme dihydropholate reductase, thus altering the synthesis of purines and pyrimidines and, lastly, on blocking viral replication through the inadequate production of tetrahydropholate. However recent findings indicate that Tropheryma whippelii’s genome lacks the encoding sequence for dihydropholate reductase (42,43) and, subsequently, the drug’s exact mechanism in bacterial metabolism has yet to be understood. On the other hand trimetoprim-sulfamethoxazole does not always prevent cerebral localization. Subsequently, 3rd generation cyclosporines are an effective alternative in this condition’s treatment.
Conclusions
Whipple’s disease is a rare complex biological disease, which is potentially lethal or anyhow highly disabling. As rare diseases must be sought to be found, specialists in internal medicine, gastroenterologists, rheumatologists, neurologists, cardiologists and physicians specialized in general medicine must be clinically highly suspicious about such a condition. The typical Whipple patient is male, aged between 50 and 70 years, with a long-standing history of migrating arthritis and signs of malabsorption, peripheral or mesenteric lymphadenomegaly and, at times, melanoderma. The clinical suspicion must be strengthened by the concurrent onset of endocardiac, thrombotic and neurological disorders.
Prof. Gino Roberto Corazza
Cattedra ed Unità di Gastroenterologia
IRCCS Policlinico San Matteo
Università di Pavia, Italia
Dr.ssa Emanuela Miceli
Cattedra ed Unità di Gastroenterologia
IRCCS Policlinico San Matteo
Università di Pavia, Italia
Bibliografia
1) Whipple GH. A hitherto undescribed disease characterized anatomically by deposits of fat and fatty acids in the intestinal and mesenteric lymphatic tissues. Johns Hopkins Hosp Bull 1907;18:382-391. www.ministerosalute.it/assistenza/malattierare - www.whipplesdisease.info
2) Allchin WH, Hebb RG. Lymphangiectasis intestini. Trans Path Soc Lond 1895;46:221-223.
3) Morgan AD. The first recorded case of Whipple’s disease? Gut 1961;2:370-372.
4) Dobbins WO III. Whipple’s disease. Springfield: Charles C Thomas, 1987.
5) Oliver Pascual E, Galan J, Oliver Pascul A, Castillo E. Un caso de lipodistrofia intestinal con lesions gangliones mesentericas de granulomatosis lipofagica. (Enfermed de Whipple) Rev Esp Enferm Aspar Digest 1947;6:213-226.
6) Farnan P. The systemic lesions of Whipple’s disease. J clin Path 1958;11:382-390.
7) Sieracki JC. Whipple’s disease; observation on systemic involvement. Arch Path 1958;66:464-467.
8) Black-Shaffer B. Tinctoral demonstration of glycoprotein in Whipple’s disease. Proc Soc Exp Biol Med 1949;72:225-227.
9) Paulley JW. A case of Whipple’s disease (intestinal lipodystrophy). Gastroenterology 1952;22:128-133.
10) Choen AS, Schimmel EM, Holt PR, Isselbacher KJ. Ultrastructural abnormalities in Whipple’s disease. Proc Soc Exp Biol Med 1960;105:411-414.
11) Silva MT, Macedo PM, Moura Nunes JF. Ultrastructure of bacilli and the bacillary origin of the macrophagic inclusions in Whipple’s disease. J Gen Microbiol 1985;131:1001-1003.
12) Relman DA, Schmidt TM, MacDermott RP, Falkow S. Identification of the uncultured bacillus of Whipple’s disease. N Engl J Med 1992;327:293-301.
13) La Scola B, Fenollar F, Fournier PE, Altwegg M, Mallet MN, Raoult D. Description of Tropheryma whipplei gen. nov., sp. nov., the Whipple’s disease bacillus. Int J Syst Evol Microbiol 2001;51(Pt 4):1471-1479.
14) Maiwald M, Schuhmacher F, Ditton HJ, Von Herbay A. Evironmental occurrence of the Whipple’s disease bacterium (Tropheryma whippelii). Appl Environ Microbiol 1998;64:760-762.
15) Ehrbar HU, Bauerfeind P, Dutly F, Koelz HR, Altwegg M. PCR-positive tests for Tropheryma whippelii in patients without Whipple’s disease. Lancet 1999;353:2214.
16) Street S, Donoghue HD, Neild GH. Tropheryma whippelii DNA in saliva of healthy people. Lancet 1999;354:1178-1179.
17) Olivieri I, Brandi G, Padula A, Di Stefano M, Mantovani W, Calabrese C, Saccoccio G, Di Febo G, Corazza GR, Miglioli M, Biasco G. Lack of association with spondyloarthritis and HLA B27 in Italian patients with Whipple’s disease. J Rheumatol 2001;28:1294-1297.
18) Bai JC, Mota AH, Maurino E, Niveloni S, Grossman F, Boerr LA, Fainboim L. Class I and Class II HLA antigens in a homogeneous Argentinian population with Whipple’s disease: lack of association with HLA B27. Am J Gastroenterol 1991;86:992-994.
19) Marth T, Neurath M, Cuccherini BA, Strober W. Defects of monocyte interleukin-12 production and humoral immunity in Whipple’s disease. Gastroenterology 1997;113:442-448.
20) Marth T, Kleen N, Stallmach A, Ring S, Aziz S, Schmidt C, Strober W, Zeitz M, Schneider T. Dysregulated peripheral ad mucosal Th1/Th2 response in Whipple’s disease. Gastroenterology 2002;123:1468-1477.
21) Corazza GR, Frisoni M, Milletti S, Vaira D, Gasbarrini G. Mechanism of thrombocytosis in Whipple’s disease. Gastroenterology 1982;82:165.
22) Nuzum CT, Sandler RS, Paulk HT. Thrombocytosis in Whipple’s disease. Gastroenterology 1981;80:1465-7.
23) Marth T, Strober W. Whipple’s disease. Semin Gastrointest Dis 1996;7:41-48.
24) Misbah SA, Ozols B, Franks A, Mapstone N. Whipple’s disease without malabsorption: new atypical features. QJM 1997;90:765-772.
25) Marth T, Roux M, Von Herbay A, Meuer SC, Feurle GE. Persistent reduction of complement receptor 3 alpha-chain expressing mononuclear blood cells and transient inhibitory serum factors in Whipple’s disease. Clin Immunol Immunopathol 1994;72:217-226.
26) Di Stefano M, Jorizzo RA, Brusco G, Cecchetti L, Sciarra G, Loperfido S, Brandi G, Gasbarrini G, Corazza GR. Bone mass and metabolism in Whipple’s disease: the role of hypogonadism. Scand J Gastroenterol. 1998 Nov;33(11):1180-5
27) Hinrikson HP, Dutly F, Altwegg M. Homogeneity of 16S-23S ribosomal intergenic spacer regions of Tropheryma Whippelii in Swiss patients with Whipple’s disease. J Clin Microbiol 1999;37:152-156.
28) Hinrikson HP, Dutly F, Nair S, Altwegg M. Detection of three different types of Tropheryma whippelii directly from clinical specimens by sequencing, single-strand conformation polymorphism (SSCP) analysis and type-specific PCR of their 16S-23S ribosomal intergenic spacer region. Int J Sysr Bacteriol 1999;49:1701-1706.
29) Maiwald M, Von Herbay A, Lepp PW, Relman DA. Organization, structure, and variability of the rRNA operon of the Whipple’s disease bacterium (Tropheryma whippelii). J Bacteriol 2000;182:3292-3297).
30) Geissdorfer W, Wittmann I, Rollinghoff M, Schoerner C, Bogdan C. Detection of a new 16S-23S rRNA spacer sequence variant (type 7) of Tropheryma whippelii in a patient with prosthetic aortic valve endocarditis. Eur J Clin Microbiol Infect Dis 2001;20:762-763.
31) Dutly F, Hinrikson HP, Seidel T, Morgenegg S, Altwegg M, Bauerfeind P. Tropheryma whippelii DNA in saliva of patients without Whipple’s disease. Infection 2000;28:219-222.
32) Gubler JG, Kuster M, Dutly F, Bannwart F, Krause M, Vogelin HP, Garzoli G, Altwegg M. Whipple endocarditis without overt gastrointestinal disease: report of four cases. Ann Intern Med 1999;131:112-116.
33) O’Duffy JD, Griffino WL, Li CY, Abdelmalek MF, Persing DH. Whipples arthritis. Direct detection of Tropheryma Whippelii in synovial fluid and tissue. Arthritis Rheum 1999;42:812-817.
34) Romanul FCA, Radvany J, Rosales RK. Whipple’s disease confined to the brain: a case studied clinically and pathologically. J Neurol Neurosurg psychiatry 1977;40:901-909.
35) Adams M, Rhyner PA, Day J, DeArmond S. Smuckleer E. Whipple’s disease confined to the central nervous system. Ann Neurol 1987;21:104-108.
36) Feurle GE, Marth T. An evaluation of antimicrobial treatment for Whipple’s disease: tetracycline versus trimethoprim-sulfamethoxazole. Dig Dis Sci 1994;39:1642-48.
37) Keinath RD, Merrell DE, Vlietstra R, Dobbins WO III. Antibiotic treatment and relapse in Whipple’s disease. Gastroenterology 1985;88:1867-73.
38) Cooper GS, Blades EW, Remler BF, Salata RA, Bennert KW, Jacobs GH. Central nervous system Whipple’s disease: relapse during therapy with trimethoprim-sulfamethoxazole and remission with cefixime. Gastroenterology 1994;106:782-786.
39) Bentley SD, Maiwald M, Murphy LD, Pallen MJ, Yeats CA, Dover LG, Norbertczak HT, Besra GS, Quail MA, Harris DE, Von Herbay A, Goble A, Rutter S, Squares R, Squares S, Barrell BG, Parkhill J, Relman DA. Sequencing and analysis of the genome of the Whipple’s disease bacterium Tropheryma whipplei. Lancet 2003;361:637-644.
40) Schaffner A, Schneemann M. Whipple’s disease: from enigma to genomics. Lancet 2003;361:632.